9262009 William Henry was an English physician and chemist who in 1803 proposed what is now called Henrys law which states that At a constant temperature the amount of a given gas dissolved in a given type and volume of liquid is directly proportional to the partial pressure of that gas in equilibrium with that liquid. 462019 Henrys law and Daltons law are central to understanding decompression sickness.
Decompression sickness or the bends.

Decompression sickness henry's law. Henrys law states that at a constant temperature the amount of a gas that is dissolved in a liquid is directly proportional to the partial pressure of that gas. Henrys Law states that the amount of a particular gas dissolved in solution is proportional to the partial pressure of that gas over the solution. Decompression sickness is caused by small nitrogen bubbles forming in the blood and tissues of the body.
Is related more to Henrys Law which states that more gas will be dissolved in a liquid when the gas is pressurized. Decompression sickness is prevented by limiting time under pressure and cured by time in a recompression chamber. As temperature increases the solubility of gases decrease in aqueous solutions.
Example-The Henrys Law constant for the solubility of nitrogen in water is 640x10-4 Matm at 250 Celsius. This phenomenon together with Daltons law helps us understand that as this happens the partial pressure of the air inside the lungs increases. At 750 atm how many grams of N 2 can be dissolved in 250 L of water at 250 degrees Celsius.
Decompression sickness is caused by nitrogen coming out of solution when a diver ascends from a dive. 1222021 Henrys law states that the solubility of a gas increases when the pressure increases. Daltons Law states that the pressure of a gas is the sum of the partial pressures of all gases.
In recreational diving limiting underwater time and depth minimizes nitrogen saturation. Henrys constant will have the units of Matm for this equation. Henrys Law shows that a gas will dissolve in a liquid in proportion to the partial pressure of that gas.
Please see below with regards to the bends and Boyles Law. In commercial diving divers are monitored carefully underwater then enter a recompression chamber when they come to the surface. What should you not do after scuba diving 11 must NOT dos after diving.
This law helps to explain the principles behind diving-related barotrauma and air embolism. Once the no-decompression limit has been passed one or more decompression stops are required during ascent to allow delayed diffusion of nitrogen out of the lipid tissues back into the blood. Answer-S 640 x 10-4 Matm0750 atm 48 x 10-4 M.
102630 Henrys Law states that the solubility of a gas in a liquid is proportional to the partial pressure of the gas over the liquid. Henrys law states that at a given temperature the amount of gas that will dissolve in a liquid is directly proportional to the partial pressure of the gas. Taking a hot bath after diving may result in decompression sickness.
If the pressure doubles the amount of gas the. Henrys gas law also states that the gass solubility is inversely proportional to the temperature. So to avoid decompression sickness a diver must monitor his depth and time to limit the amount of nitrogen on-gassing and he must ascend slowly enough that the pressure is released slow enough to allow the nitrogen to leave the tissues without forming bubbles.
Because of the water pressure body tissue absorbs nitrogen gas faster as a diver descends than when ascending to the surface. In organic solutions the solubility of gases increases at higher temperatures. This law provides the explanation for decompression sickness and nitrogen narcosis.
The inert gas that evolves from the tissues can then cause a mass effect and inflammatory response in that tissue. DCS is discussed in more detail in the Diving Injuries section. 4222013 This gas law now appropriately known as Henrys Law together with Boyles Law form the basis of the pathophysiology and treatment for Decompression Illness.
7122020 Henrys Law and Freediving When you descend on a dive according to Boyles law the increase in pressure leads to a decrease of the volume of air in your lungs. 352019 According to the Henry law once nitrogen has saturated tissues a standard ascent to the surface with decreasing ambient pressure can allow nitrogen to bubble out of solution. An everyday example is given by ones experience with carbonated soft drinks which contain dissolved carbon dioxide.
An example where Henrys law is at play is in the depth-dependent dissolution of oxygen and nitrogen in the blood of underwater divers that changes during decompression leading to decompression sickness. This comes about as a result of the reducing air pressure in the case of flying. It is sometimes described as being caused by evolved gas.
1292021 Simply put the more pressure there is the more gas can dissolve and blend into the liquid. If there is an increase in temperature the gass solubility decreases. Decompression Sickness and Henrys Law animation This video file cannot be played.
As pressure rises the amount of nitrogen that the tissues will hold also rises. When diver surfaces slowly heshe will have a reduced impact of pain from the bubbles that form.
Effects Of Increased Dissolved Nitrogen From Scuba Diving Decompression Sickness
 Diffusion And Decompression Sickness Divers Who Want To Learn More
Diffusion And Decompression Sickness Divers Who Want To Learn More
 Marine Vertebrates Lecture 12 Diving Part 2 Part 2 Diving Physiology Diving Depth Records Humans Free Diving Mandy Cruickshank 78 M Martin Ppt Download
Marine Vertebrates Lecture 12 Diving Part 2 Part 2 Diving Physiology Diving Depth Records Humans Free Diving Mandy Cruickshank 78 M Martin Ppt Download
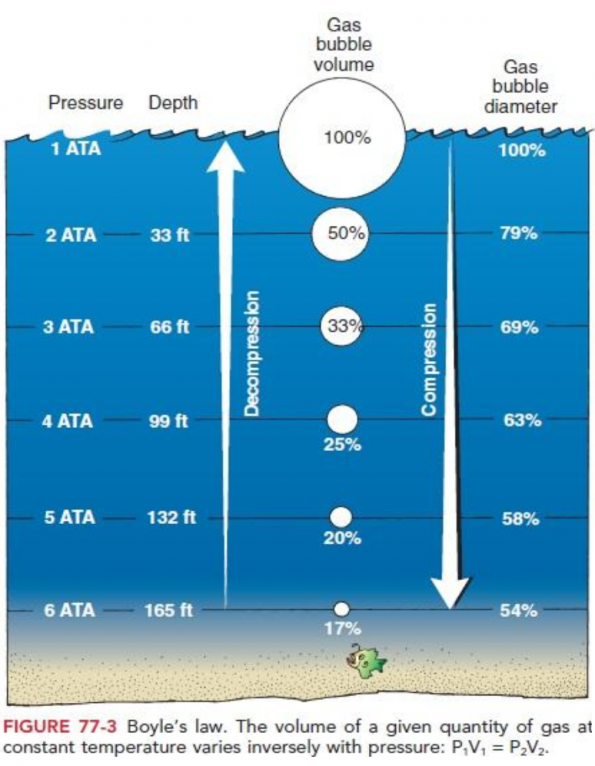 Fun With Bubbles Dysbarism And Hyperbaric Oxygen Therapy Emottawa
Fun With Bubbles Dysbarism And Hyperbaric Oxygen Therapy Emottawa
 What Does Henry S Law Mean In Diving Rateyourdive Com
What Does Henry S Law Mean In Diving Rateyourdive Com
Effects Of Increased Dissolved Nitrogen From Scuba Diving Decompression Sickness
 Ppt Decompression Sickness Powerpoint Presentation Free Download Id 1815000
Ppt Decompression Sickness Powerpoint Presentation Free Download Id 1815000
 Henry S Law In Regards To Ascents The Merbabe Adventures
Henry S Law In Regards To Ascents The Merbabe Adventures
What You Need To Know About Gas Mindmap Voorbeeld
 Decompression Illness Rcemlearning
Decompression Illness Rcemlearning
 Powerschool Learning Altitude And Transport Physiology The Gas Laws Effects On You Your Crew And Your Patients
Powerschool Learning Altitude And Transport Physiology The Gas Laws Effects On You Your Crew And Your Patients
 Henry S Law And The Bends In Scuba Diving By Jesse Collins
Henry S Law And The Bends In Scuba Diving By Jesse Collins
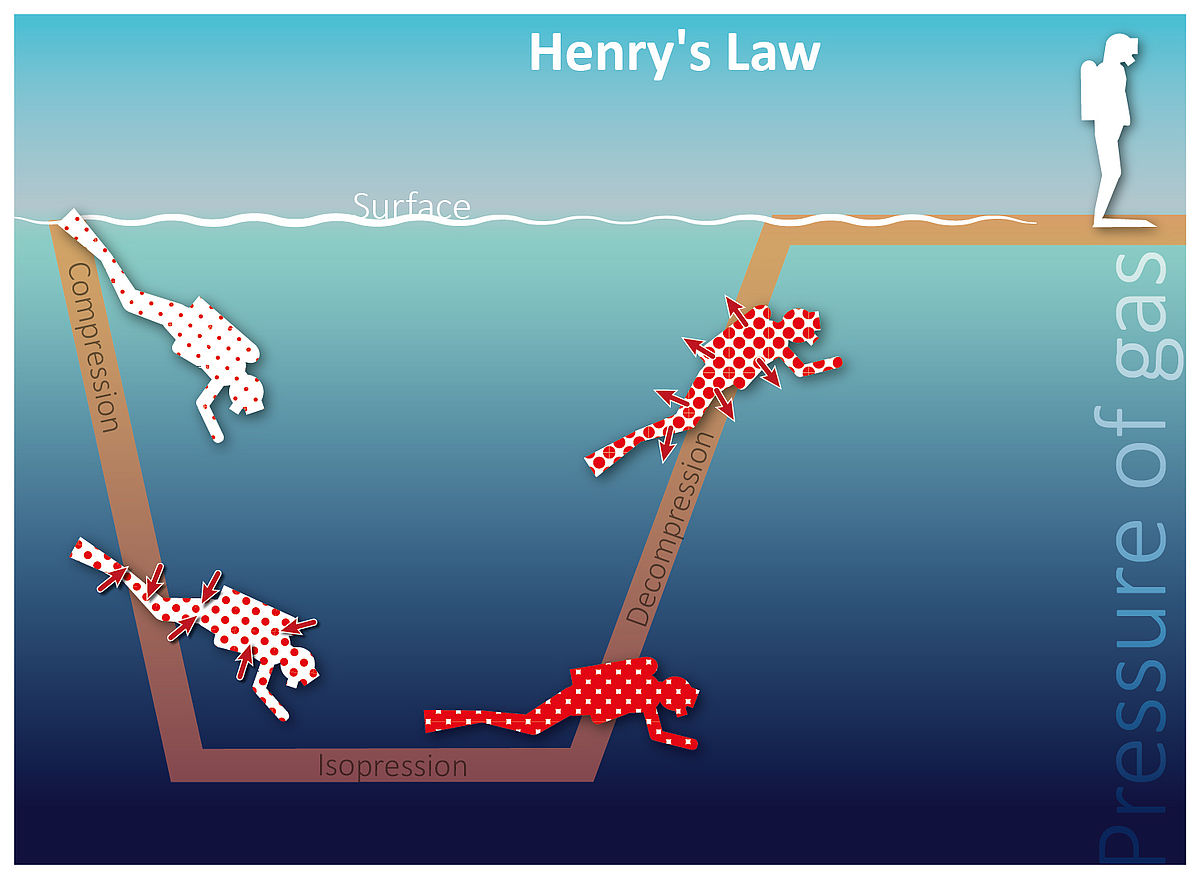 Decompression Sickness Medical Articles Service Medicine Aqua Med
Decompression Sickness Medical Articles Service Medicine Aqua Med
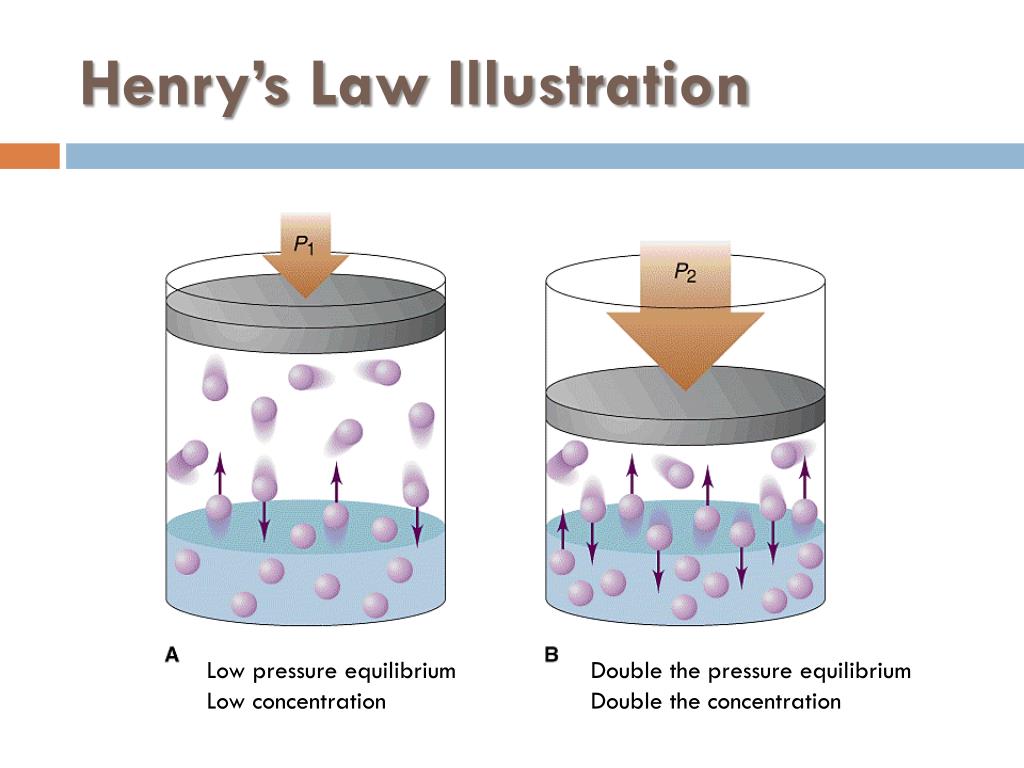 Ppt Decompression Sickness Powerpoint Presentation Free Download Id 1815000
Ppt Decompression Sickness Powerpoint Presentation Free Download Id 1815000






Post a Comment for "Decompression Sickness Henry's Law"